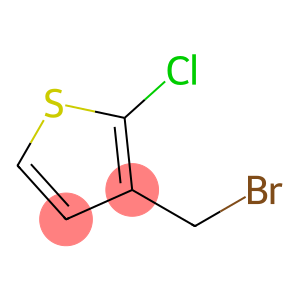
Thiophene, 3-(bromomethyl)-2-chloro-
2-Chloro-3-bromomethylthiophene
CAS: 40032-81-3
Molecular Formula: C5H4BrClS
Thiophene, 3-(bromomethyl)-2-chloro- - Names and Identifiers
| Name | 2-Chloro-3-bromomethylthiophene |
| Synonyms | 3-bromomethyl-2-chlorothiophene 2-Chloro-3-bromomethylthiophene 2-CHLORO-3-BROMOMETHYLTHIOPHENE 3-(Bromomethyl)-2-chlorothiophene thiophene, 3-(bromomethyl)-2-chloro- Thiophene, 3-(bromomethyl)-2-chloro- |
| CAS | 40032-81-3 |
| InChI | InChI=1/C5H4BrClS/c6-3-4-1-2-8-5(4)7/h1-2H,3H2 |
Thiophene, 3-(bromomethyl)-2-chloro- - Physico-chemical Properties
| Molecular Formula | C5H4BrClS |
| Molar Mass | 211.51 |
| Density | 1.757±0.06 g/cm3(Predicted) |
| Boling Point | 88 °C(Press: 1 Torr) |
| Flash Point | 89.914°C |
| Vapor Presure | 0.132mmHg at 25°C |
| Storage Condition | under inert gas (nitrogen or Argon) at 2–8 °C |
| Refractive Index | 1.618 |
Thiophene, 3-(bromomethyl)-2-chloro- - Introduction
It is an organic compound containing chlorine and bromine functional groups. Its molecular formula is C5H3BrClS and its molecular weight is 215.50g/mol. The following is a detailed description of the properties, uses, methods of manufacture and safety information:
Nature:
-Appearance: It is a colorless to yellowish liquid or crystalline solid.
-Melting point: about 25-28 degrees Celsius.
-Boiling point: about 180-185 degrees Celsius.
-Soluble: Soluble in organic solvents, such as methanol, dichloromethane, etc.
Use:
-is often used as an intermediate in organic synthesis, and can be used to synthesize drugs, pesticides, photosensitive materials and other compounds with thiophene skeleton.
-It is also used as an organic semiconductor material in optoelectronic devices, such as organic solar cells, organic field effect transistors, etc.
Preparation Method:
There are many preparation methods, and the following is one of the common methods:
2-methylthiophene is reacted with tribromomethane to generate 2-bromo-3-methylthiophene. The 2-bromo-3-methylthiophene is then reacted with hydrogen chloride to give the final product, No.
Safety Information:
-There are few specific hazards and toxicity data, so it is necessary to take appropriate safety measures when using it, such as wearing protective gloves, goggles and protective clothing.
-During operation, direct contact with skin and eyes should be avoided. After use should be thoroughly cleaned, and maintain good ventilation conditions.
-It may be toxic to aquatic organisms, and care should be taken to avoid its entry into water sources and the environment.
-During storage and transportation, avoid contact with oxidants, strong acids, strong bases and other substances to prevent dangerous reactions.
Nature:
-Appearance: It is a colorless to yellowish liquid or crystalline solid.
-Melting point: about 25-28 degrees Celsius.
-Boiling point: about 180-185 degrees Celsius.
-Soluble: Soluble in organic solvents, such as methanol, dichloromethane, etc.
Use:
-is often used as an intermediate in organic synthesis, and can be used to synthesize drugs, pesticides, photosensitive materials and other compounds with thiophene skeleton.
-It is also used as an organic semiconductor material in optoelectronic devices, such as organic solar cells, organic field effect transistors, etc.
Preparation Method:
There are many preparation methods, and the following is one of the common methods:
2-methylthiophene is reacted with tribromomethane to generate 2-bromo-3-methylthiophene. The 2-bromo-3-methylthiophene is then reacted with hydrogen chloride to give the final product, No.
Safety Information:
-There are few specific hazards and toxicity data, so it is necessary to take appropriate safety measures when using it, such as wearing protective gloves, goggles and protective clothing.
-During operation, direct contact with skin and eyes should be avoided. After use should be thoroughly cleaned, and maintain good ventilation conditions.
-It may be toxic to aquatic organisms, and care should be taken to avoid its entry into water sources and the environment.
-During storage and transportation, avoid contact with oxidants, strong acids, strong bases and other substances to prevent dangerous reactions.
Last Update:2024-04-09 20:49:11
Supplier List
Spot supply
Product Name: 2-Chloro-3-bromomethylthiophene Visit Supplier Webpage Request for quotationCAS: 40032-81-3
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: 2-Chloro-3-bromomethylthiophene Request for quotation
CAS: 40032-81-3
Tel: +86 13001858685
Email: 13001858685@163.com
QQ: 1731087008
WhatsApp: +86 13001858685
CAS: 40032-81-3
Tel: +86 13001858685
Email: 13001858685@163.com
QQ: 1731087008
WhatsApp: +86 13001858685
Product Name: 2-Chloro-3-bromomethylthiophene Visit Supplier Webpage Request for quotation
CAS: 40032-81-3
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 40032-81-3
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
Spot supply
Product Name: 2-Chloro-3-bromomethylthiophene Visit Supplier Webpage Request for quotationCAS: 40032-81-3
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: 2-Chloro-3-bromomethylthiophene Request for quotation
CAS: 40032-81-3
Tel: +86 13001858685
Email: 13001858685@163.com
QQ: 1731087008
WhatsApp: +86 13001858685
CAS: 40032-81-3
Tel: +86 13001858685
Email: 13001858685@163.com
QQ: 1731087008
WhatsApp: +86 13001858685
Product Name: 2-Chloro-3-bromomethylthiophene Visit Supplier Webpage Request for quotation
CAS: 40032-81-3
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 40032-81-3
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
View History


